Everyone recognizes a diamond when they see one. Mounted on a ring or embedded in a necklace, these gemstones are prized for their use in jewelry around the world. What many people don’t realize, however, is that diamonds offer much more than commercial value.

While the use of diamonds has been traced back to ancient times, large-scale modern diamond mining began in the late 1860s with the discovery of diamonds in Kimberly, South Africa. The rock from which the diamonds were extracted was creatively named kimberlite, after the town. Today, kimberlite is by far the most important source of naturally occurring diamonds, and deposits are found around the world.

Diamonds originate from deep in the mantle, hundreds of kilometres beneath Earth’s surface. A common misconception is that diamonds are formed through the metamorphism of coal. This is not the case – although they are both composed of carbon, coal deposits are a result of the burial of plants, whereas diamonds are much older than the first plants on Earth! The journey of a diamond from the mantle to the surface is fascinating, and to properly set the stage, we need to first learn about plate tectonics.
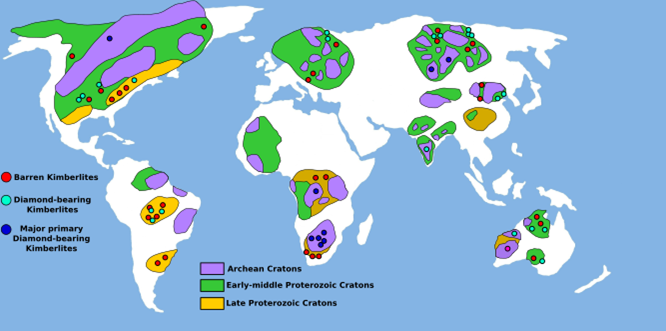
Large, rigid continental plates move slowly on Earth’s surface, repeatedly reshaping our continents over millions of years. In some parts of the continental lithosphere, there are massive and ancient blocks that haven’t been a part of subduction or continental rifting processes in billions of years. These are called cratons, and they form the geologically stable interiors of present-day continents. Cratons are found on all continents, including North America; for example, the Superior Province extends across Ontario and Manitoba and forms the core of the Canadian Shield. Typically, cratons are older than 2.5 billion years, meaning they come from a geological eon called the Archean. These cratons are thick enough to reach into the mantle, creating the perfect environment for diamonds to grow – anywhere else, and the pressures and temperatures are not suitable for the crystallization of carbon atoms in diamond form. It’s on the surface of these ancient cratons that diamond-rich kimberlites are found, but the kimberlites themselves are significantly younger than their Archean hosts; most are younger than 150 million years old. So how did they get there?
Transporting a diamond from beneath an Archean craton to the surface of Earth is no easy feat, not least of all because the conditions of Earth’s surface are far below a diamond’s “comfort zone”, so to speak. If you were to bring a diamond to the surface at normal geological speeds, it would convert to graphite, the physical arrangement of carbon atoms that is stable at normal pressure and temperature conditions. To keep its diamond passengers happy, the magma must follow a few rules.
Kimberlite magmas are full of volatiles, like water and carbon dioxide, and are more magnesium-rich than typical magmas. This type of chemistry produces low-density, low-viscosity magma that wants to separate from the mantle and rise to the surface in a truly remarkable way. Geological models show that kimberlitic magma begins to move upward at speeds ranging from 4–20 metres per second, propelled to even faster speeds by decreasing pressures towards an eventual 200 m/s explosive eruption on the surface. Assuming a starting depth of about 300 kilometres below the surface, a kimberlite could complete its diamond delivery within 15 hours! Considering that many geological features occur over millions of years, kimberlites move from the mantle to the surface relatively instantaneously. The combination of the unique volatile-rich magma chemistry and rapid ascent help to keep the diamonds stable, so they don’t convert to graphite or dissolve into the magma. This eruptive transport gives kimberlite pipes their characteristic “carrot” shape that is sometimes accompanied by a fallback of ejected pyroclastic material, like ash and rock fragments.
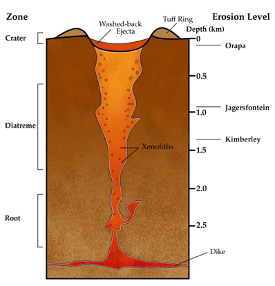
Since diamonds do not crystallize within the original kimberlite magma and are instead picked up from the mantle as the magma ascends, they’re called xenocrysts, or foreign crystals. But while diamonds tend to get most of the attention, kimberlites can also carry other mantle rock fragments (xenoliths) to the surface. In fact, diamonds themselves often contain mineral inclusions, and while these are heartlessly referred to as flaws by a jeweller, they can be immensely important for understanding the pressure and temperature conditions of diamond growth. So, not only are kimberlites an important source of economic geology, but they can also provide geologists with information about the chemistry and conditions of the deep mantle that we would otherwise be unable to observe. Sadly, there hasn’t been much kimberlite activity in the last 50 million years, which means that the diamonds on our surface today are likely the only ones we’ll ever find on Earth.

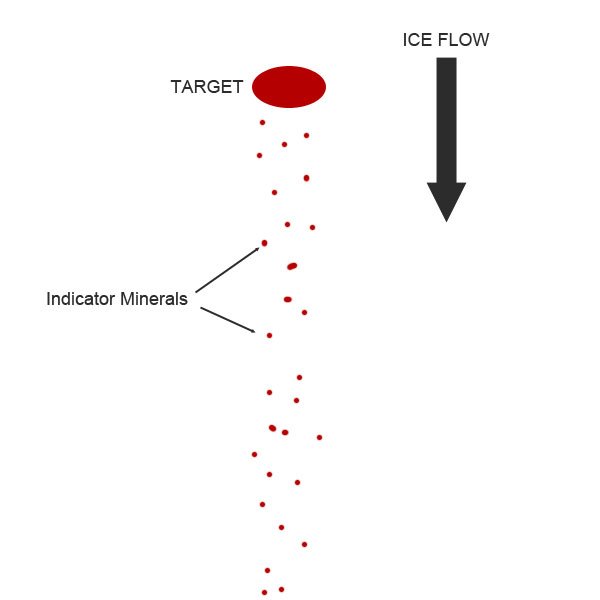
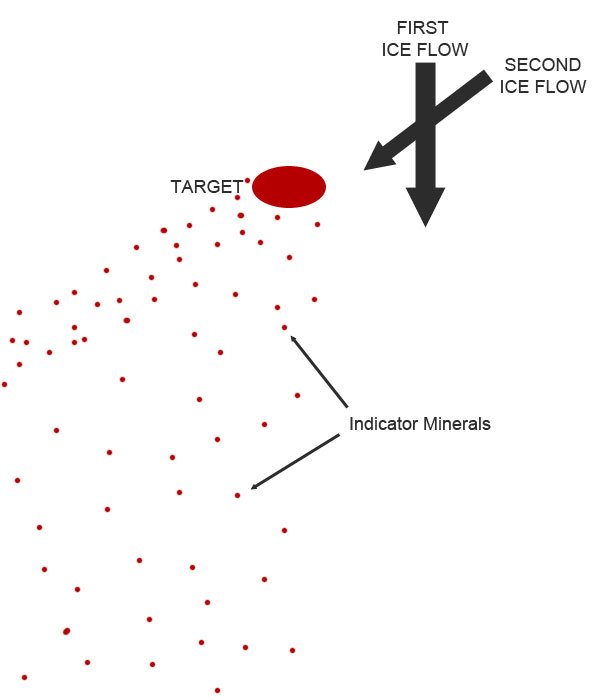
Even though kimberlite deposits occur on all continents, they can be somewhat difficult to track down. Rocks from the mantle tend to be unstable on the surface of Earth, so even though kimberlites are much younger than their surrounding rock, their composition makes them more vulnerable to weathering and erosion processes. However, specific minerals in kimberlites, like garnet and ilmenite, can survive weathering and in the case of many deposits in Canada, glacial transport. When the kimberlite deposit is hit by an advancing glacier, resistant minerals from the kimberlite are carried along by the ice and exploration geologists can follow the path of these minerals against the direction of glacier transport to find the source kimberlite pipe. Indicator minerals are abundant in kimberlites and relatively rare in other igneous rocks, making them a very useful tool for finding a kimberlite pipe that today might be a crater filled by water or vegetation. This method, also known as drift prospecting, is widely used across Ontario and the northern territories to search for diamond deposits because of Canada’s extensive glacial history.
While diamonds are relatively rare and difficult to find on Earth, they’re actually exceedingly common in space. However, at a billionth of a meter in diameter, they’re nowhere near big enough for an engagement ring. These nanodiamonds are abundant in interstellar dust and nebulae and were first identified in meteorites that pre-date the solar system. On the icy surfaces of the gas giants Uranus and Neptune, diamonds may be able to crystallize from high-pressure methane gas in the. Astronomers have even identified exoplanets in star systems that, based on the amount of carbon their suns produce, would be carbon-based rather than silicon-based, as is our own rocky planetary neighbourhood. Carbon planets with a high enough density to be crystalline would therefore be composed entirely of diamond.
To many geologists, the true value of a diamond is not in its weight, cut, or clarity. A diamond is a window into the unfathomable depths of the deep mantle and can provide exceptional information about the composition and conditions of Earth’s interior. They tell a fascinating story of an expedition across many aspects of geoscience; billion-year-old crystallization below ancient cratons, an explosive volcanic ascent to the surface, finished by glacial dispersion and sedimentary processes. While many people recognize the immense commercial value of a diamond, perhaps their true contribution is the valuable insight they provide into the formation of Earth, the distant past, and even the far reaches of our universe.

Zachary Adam is a graduate student at the University of Waterloo, pursuing his Master of Science degree in Geology. His current research focuses on understanding controls on gold mineralization in Archean greenstone belts. He has a keen interest in structural geology, with a focus on mineral deposits and plate tectonics, and has spent many seasons in the field working on gold and diamond exploration projects around Ontario.

